Oral solid dosage forms remain the most widely adopted drug delivery systems due to their convenience, stability, and patient compliance. However, for drugs with site-specific absorption in the upper gastrointestinal (GI) tract, rapid gastric emptying often limits bioavailability.
To address this challenge, bilayer gastroretentive modified-release (GR-MR) tablets have emerged as a sophisticated formulation strategy. By integrating an immediate-release (IR) layer for rapid onset and a gastroretentive extended-release (GR-ER) layer for prolonged gastric residence, this system enables both precise release kinetics and enhanced in vivo exposure.
This white paper addresses a critical and often overlooked aspect of product development by applying a Quality-by-Design (QbD) framework to design and control product quality.

Figure 1: Bilayer tablets
Gastroretentive drug delivery systems (GRDDS) can be broadly categorized as follows:
Table 1. Overview of GRDDS Technologies: mechanisms, advantages and limitations
Category | Subtype | Mechanism | Advantages | Limitations |
GRDDS | Floating | Low density or gas generation enables buoyancy | Potential retention; improved absorption | Sensitive to gastric conditions |
Swelling / Expandable | Polymer swelling to size > pylorus | Reliable retention | Requires strong mechanical integrity | |
Mucoadhesive | Adhesion to gastric mucosa | Enhanced absorption | Variable in vivo adhesion | |
High-Density | Density >2.5 g/cm³ for retention | Resists emptying | Limited practicality | |
Raft System | Gel raft formation | Localized retention | Environment-dependent | |
Magnetic | External magnetic control | Precise (theoretical) | Not clinically practical |
Among these, swelling systems are most commonly used in bilayer designs due to their balance of feasibility and performance.
Table 2. Comparison of different oral drug delivery systems
System | Key Advantage | Limitation |
IR | Rapid onset | Short duration |
ER | Sustained exposure | Delayed onset |
GRDDS | Enhanced upper GI absorption | In vivo variability |
Bilayer IR + GR-ER | Immediate + prolonged + localized exposure | High complexity |
This bilayer design is particularly valuable for:
· Narrow absorption window drugs
· Molecules with solubility-limited absorption
· Therapies requiring rapid onset + sustained exposure
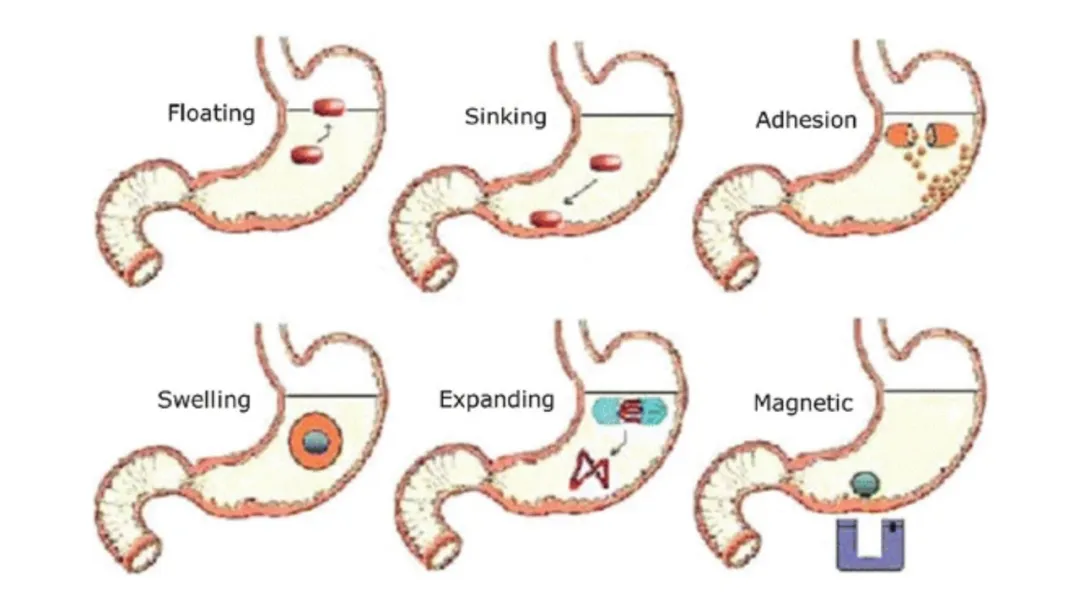
Figure 2: Mechanisms of Action of Various Gastroretentive Tablets
1. Quality Challenges: Where Complexity Emerges
Bilayer GR tablets introduce multi-dimensional quality risks:
Structural risks
· Interlayer adhesion failure
· Delamination during handling or dissolution
Performance risks
· Uncontrolled swelling or erosion
· Inconsistent retention behavior
· Dual-release profile variability
Manufacturing risks
· Compression sequence sensitivity
· Layer weight variation
· Polymer hydration kinetics
These complexities make QbD not optional—but essential.
2. QTPP → CQAs: Translating Clinical Goals into Quality Targets
Quality Target Product Profile (QTPP)
· Rapid onset (IR layer)
· Controlled release (8–24 h)
· Reliable gastric retention
· Mechanical robustness during swelling
· Stable performance across shelf life
These translate into the following Critical Quality Attributes (CQAs).
3. Critical Quality Attributes (CQAs)
(1) Bilayer Integrity & Mechanical Strength
· Adhesion strength between layers
· Tablet hardness / friability
· Resistance to delamination
Crystal Pharmatech incorporate compaction simulation to provide comprehensive mechanical properties for both layer composition powders and implement a special testing device to measure layer-layer bonding strength to guide bilayer tablet formulation development.

Figure 3: STYL′One Compaction Simulator
(2) Swelling & Matrix Expansion Behavior
Driven by hydrophilic polymers (e.g., HPMC, PEO):
· Swelling index
· Matrix integrity
· Expansion kinetics
Mature in-vitro swelling index evaluation methodology in CP
(3) Layer-Specific Dissolution
· IR+ER dose combination design strategy by PBPK modeling
· IR: rapid release (e.g, >80% in ~30 min)
· ER: controlled release over hours
4. Critical Material Attributes (CMAs)
Polymer-related
· HPMC viscosity grade
· PEO molecular weight
· Hydration rate
API-related
· Particle size
· Solubility / pKa
· Solid-state form
M3(Material+Molecule+Medicine) philosophy to fully understand the material and molecule physiochemical properties to select the desired excipients for formulation composition.
5. Critical Process Parameters (CPPs)
Compression process
· First-layer pre-compression force
· Main compression force
· dwell time
Layer control
· Weight ratio (IR vs ER)
Granulation
· Binder level
· Residual moisture
Using advanced compaction simulations to predict mechanical behavior, layer adhesion, and compression profiles, etc. Reduces trial-and-error, and accelerates development of robust, high-quality oral dosage forms.
All CPPs should be mapped to CQAs via DoE-based design space.
6. Dissolution & In Vitro Performance Testing
Dissolution is the core quality control tool:
Method design
· Medium: simulated gastric fluid (pH 1.2)
· Duration: up to 24 h
· Apparatus: paddle (USP II)
Advanced characterization
· Swelling kinetics
· Matrix imaging
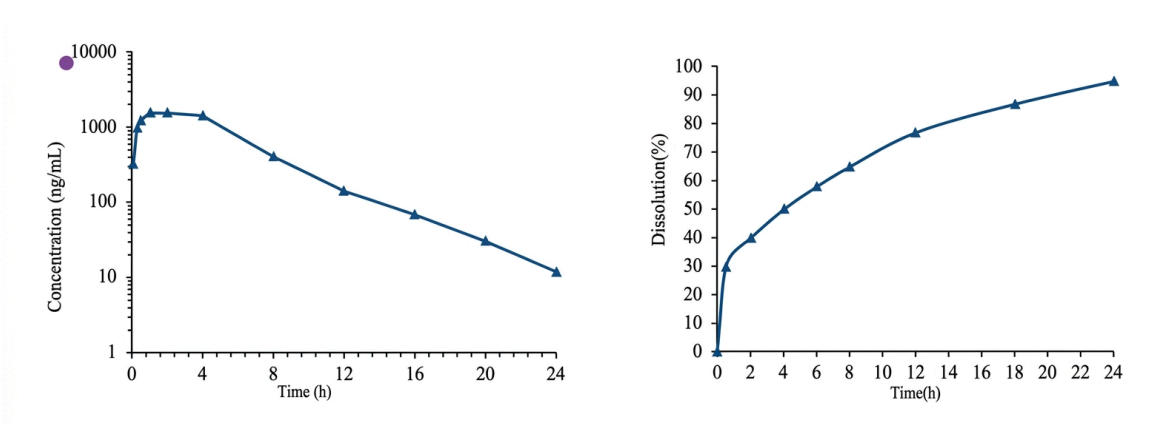
Figure 4: PK data and in vitro dissolution of gastric-retentive bilayer sustainedrelease tablets (simulated curves from GastroPlus)
7. Stability
Key degradation risks
Table 3. Degradation mechanisms and corresponding mitigation strategies
Degradation Mechanism | Main Source | Affected API Structures | Mitigation Strategies |
Oxidative degradation | PEO oxidation products, process or residual PEO | Phenols, amines, thioethers, heterocycles | Add antioxidants, use low-peroxide PEO |
Acid-catalyzed degradation | Acids generated from PEO oxidation | Esters, amides, hydrolysis-sensitive groups | Control local pH, add buffers |
Metal-catalyzed oxidation | PEO adsorbed metal impurities | Phenolic, aromatic amine, easily oxidized structures | Add chelating agents, control metal content |
8. A Practical QbD Framework
A robust development strategy typically follows:
1. Define QTPP
2. Identify CQAs
3. Link CMAs & CPPs via risk assessment
4. Apply DoE to establish design space
5. Develop discriminatory dissolution methods
6. Confirm robustness via stability + scale-up
Developing bilayer gastroretentive MR tablets is far more than combining two functional layers. It requires:
· Deep understanding of material science
· Precise control of process parameters
· Systematic application of QbD principles
When executed effectively, this platform enables:
✔ Precise release control
✔ Prolonged gastric retention
✔ Enhanced bioavailability for challenging molecules
Crystal Pharmatech, founded in 2010, is a global contract research organization with approximately 300 employees and four R&D centers in New Jersey (USA), San Francisco (USA), Toronto (Canada), and Suzhou (China). Collectively, these sites provide integrated support for pharmaceutical and biotechnology companies worldwide. Our capabilities span three specialized platforms: in small molecules, we offer API solid state research and crystallization, preformulation, formulation development, and GMP manufacturing and supply; under Crystal Bio Solutions, we deliver bioanalytical and biomarker testing, biologics characterization and CMC analytics, and clinical pharmacology; and through Crystal NAX (Nucleic Acid Excellence), we provide end-toend solutions for nucleic acid therapeutics, from early research through clinical development.
[1] Khar,R.K., Vyas,S.P.,Ahmad,F.J.,& Jain,G.K.(2013).Gastroretentive Drug Delivery Systems.CRC Press.https://doi.org/10.1201/b14636
[2] Singh,B.N.,& Kim,K.H.(2000).Gastroretentive drug delivery systems:An approach to oral controlled drug delivery via gastric retention. Journal of Controlled Release,63(3),235-259.https://doi.org/10.1016/S0168-3659(99)00204-7
[3] Arora,S., Ali,J.,Ahuja,A.,Khar,R.K.,& Baboota,S.(2005).Floating drug delivery systems: A review.AAPS PharmSciTech,6(3),E372-E390. https://doi.org/10.1208/pt060347
[4] Talukder,R.,& Fassihi,R.(2004). Gastroretentive delivery systems: A mini-review.Drug Development and Industrial Pharmacy,30(10),1019-1028. https://doi.org/10.1081/DDC-200033481
[5] Bardonnet,P.L.,Faivre,V.,Piffaretti,J.C.,& Falson,F.(2006). Gastroretentive dosage forms: Overview and special case of helicobacter pylori.International Journal of Pharmaceutics, 277(1-2),1-13. https://doi.org/10.1016/j.ijpharm.2005.12.029
[6] Streubel,A., Siepmann,J.,& Bodmeier,R.(2003). Floating matrix tablets based on low density foam powder: Effects of formulation and processing parameters on drug release. European Journal of Pharmaceutics and Biopharmaceutics,55(3),329-335.https://doi.org/10.1016/S0939-6411(02)00171-2
[7] Prajapati, V.B.,Jani,G.K.,Moradiya,N.G.,& Randeria,N.P.(2015).Gastroretentive drug delivery systems: A review. International Pharmaceutical Investigation,5(3),147-157.https://doi.org/10.4103/2230-973X.160848
[8] Chen,R.N.,Ho,H.O.,&Sheu,M.T.(1999). Characterization of captopril microparticle preparations and the determination of their transport across intestinal epithelial cells. International Journal of Pharmaceutics, 184(2), 161-170.https://doi.org/10.1016/S0378-5173(99)00094-2
[9] Klausner, E. A., Lavy, E., Friedman, M.,& Hoffman, A.(2003).gastroretentive dosage forms. Journal of Controlled Release, 90(2), 143-162.gastroretentive dosage forms. Journal of Controlled Release, 90(2), 143-162.https://doi.org/10.1016/S0168-3659(03)00139-7
[10] Wen, H.,& Park, K.(Eds.).(2010). Oral Controlled Release Formulation Design and Drug Delivery: Theory to Practice. Wiley.https://doi.org/10.1002/9780470537240
Subscribe to be the first to get the updates!