Mol2Med™ First-Time-Right Approach for Small Molecule Drug Development
Crystal Pharmatech is a global solid-state CRO and formulation CDMO specializing in small molecule drug development, solid form research, preformulation, formulation development, and GMP manufacturing.
Mol2Med™ is our integrated drug development platform designed to guide compounds from lead optimization to First-in-Human (FIH) studies through a data-driven, First-Time-Right strategy. By integrating developability assessment, solid-state science, crystallization development, preformulation, and formulation development, Mol2Med™ helps biotech and pharmaceutical companies accelerate programs toward clinical trials while reducing development risk.
The Fit-for-Purpose Trap
Traditional rapid-development strategies such as powder-in-capsule formulations or prototype tablets may allow compounds to enter early studies quickly. However, these approaches often fail to account for key API physicochemical properties, solid-state behavior, and manufacturability considerations. Without early integration of these factors, many programs encounter formulation failures during clinical development.
The Mol2Med™ Solution
The Mol2Med™ approach integrates developability assessment, solid-state research, crystallization development, preformulation studies, and formulation design into a unified workflow. By evaluating molecular properties, material behavior, and clinical requirements together, Mol2Med™ enables informed decision-making that supports successful progression toward First-in-Human studies.
Key Advantages
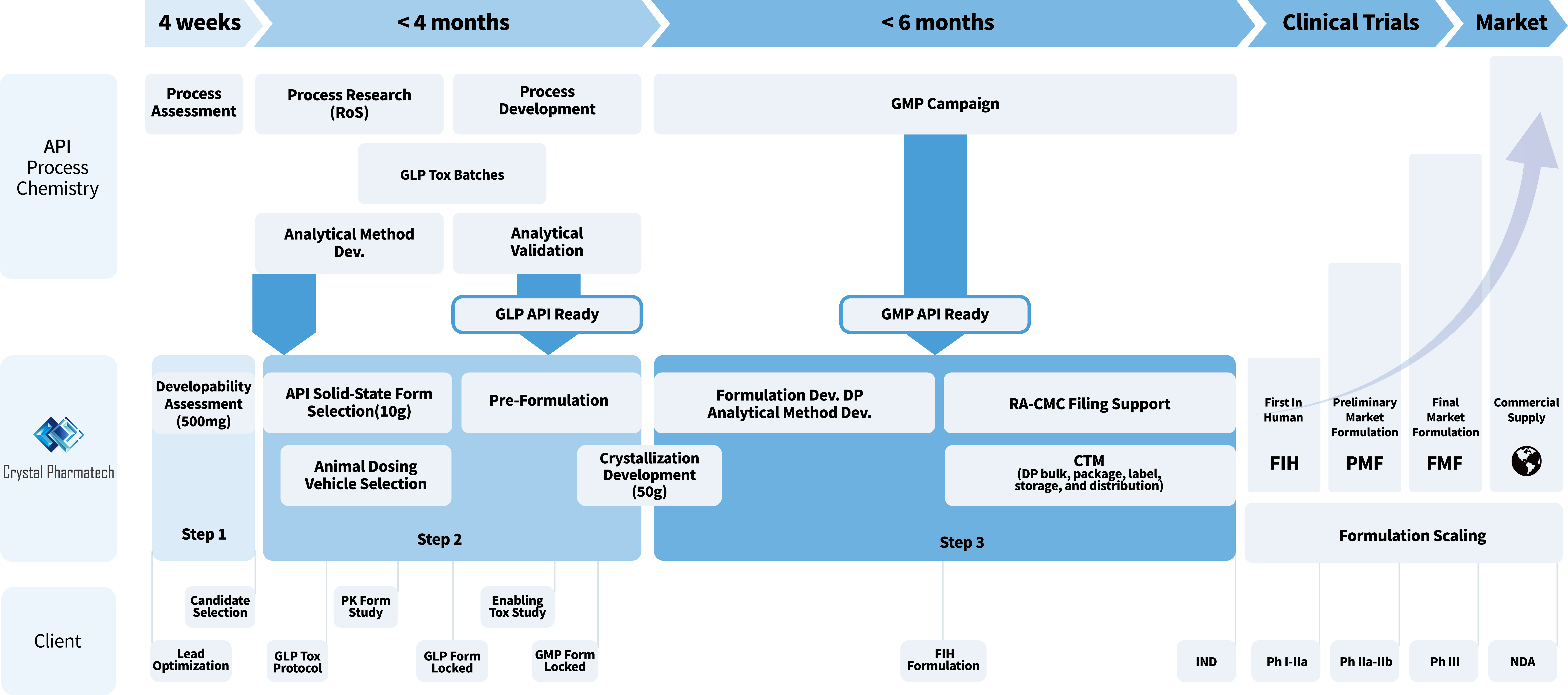
The 3-Step Framework
Developability Assessment
Our approach determines the physicochemical properties of lead candidates against industry standards to find the most "developable" lead. For a given preclinical candidate, this determines whether a free form or salt, and a conventional or enabling formulation, should be selected.
Solid Form & Pre-Formulation
This stage begins with comprehensive screening and selection of the optimal crystalline form for the API. Subsequently, we develop an optimal preclinical formulation specifically designed to support robust PK/PD and GLP Tox animal studies.
Formulation Dev & CTM Manufacturing
We execute an API-specific strategy that yields the best formulation for Phase I and clinical studies. This allows late-stage development to focus strictly on process optimization without needing significant re-formulation or human PK bridging studies.
Proven Experience in Small Molecule Development
4,000+
Drug Molecules Supported
Crystal Pharmatech supports global biotech and pharmaceutical companies advancing small molecule programs toward clinical development. The Mol2Med™ First-Time-Right approach ensures that critical CMC decisions are made early and correctly, aligning API form selection, formulation design, and manufacturing strategy.
Start Your Mol2Med™ Program
If your team is advancing a small molecule candidate, Mol2Med™ can help you move from lead optimization to clinical trials faster and with lower risk. Partner with Crystal Pharmatech to integrate discovery insights, CMC development, and clinical manufacturing into one coordinated program.
Have a question or need support with your project? Please complete the form, and our team will get back to you shortly.
Our capabilities span three specialized platforms:
Small Molecule
Crystal Bio Solutions
Crystal NAX
By providing your e-mail address, you agree to receive an e-mail response from Crystal Pharmatech to your inquiry. The information you submit will be governed by our Privacy Policy.
Subscribe to be the first to get the updates!