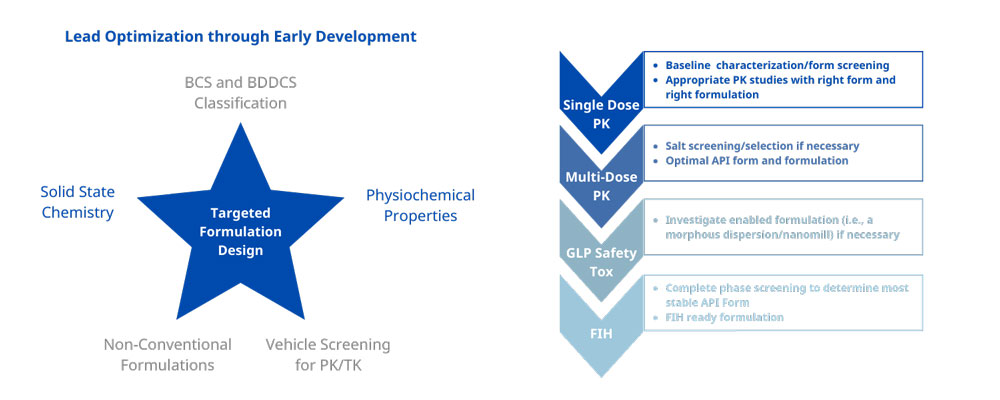
Early formulation development is a science, not trial-and-error. We utilize decision trees and 80+ years of innovative experience to deliver precise, high-performance PK studies.
| Technology | Result/Platform | Target |
|---|---|---|
| Micronization | Jet-milling | 1~10 μm |
| Nano-milling | Nano-suspension | <1000 nm |
| Spray Drying | ASD | Solubility Enhancement |
| Freeze Drying | Lyophilization | Stable Prototype |
| Extruder | Liposome | Complex Delivery |
Delivering PK/Tox-ready ASD prototypes in 2-4 weeks, not months.
API thermal assessment & solvent compatibility screening for a scientific baseline.
Film casting and dissolution testing to ensure matrix compatibility.
Utilizing precision spray drying to create stable ASD formulations.
Validated through in-vivo evaluation for performance verification.
Optimized lead ASD formulations that directly support IND-enabling studies and keep your development timelines on track.
Have a question or need support with your project? Please complete the form, and our team will get back to you shortly.
Our capabilities span three specialized platforms:
Small Molecule
Crystal Bio Solutions
Crystal NAX
By providing your e-mail address, you agree to receive an e-mail response from Crystal Pharmatech to your inquiry. The information you submit will be governed by our Privacy Policy.
Subscribe to be the first to get the updates!